1. Introduction
2. Materials and Methods
Sampling sites and sample collection
Histology
P. olseni Infection Intensity
3. Results and Discussion
1. Introduction
The Jedo Venus clam Leucoma (=Protothaca) jedoensis, (Lischke, 1874) is a common venerid clam occurring in coarse sand flat from the low intertidal to shallow subtidal along the south coast of Korea, as well as in coastal regions of Japan and China (Okutani 2000; Qi 2004; Park et al. 2006; Park and Yoon 2008). This filter-feeding clam often coexists with Manila clam Ruditapes philippinarum (Manila clam) along low-tide shorelines and is considered to be one of the vital shellfish resources on the south coast (Kim et al. 2002; Jung et al. 2004). In general, the color, type, and size of the Jedo Venus clam exhibit variation that depends on environmental parameters, including water depth, temperature, turbidity, nutrient availability, and growth duration (Park and Yoon 2008). Several studies have comprehensively assessed the reproductive cycle, age and growth, and sexual maturation of Jedo Venus clams on the south coast of Korea (Kim et al. 2002, 2003; Jo et al. 2004). According to Park et al. (2006), L. jedoensis is infected by the protozoan parasite Perkinsus olseni, which has been identified as a major pathogen responsible for the mass mortality of Manila clams and other marine mollusks (Park and Choi 2001; Villalba et al. 2004; Lee et al. 2020, 2021).
Perkinsosis has been officially designated as a notifiable disease by the World Organization for Animal Health (OIE), leading to international measures for its regulation and control (OIE 2021). As determined through DNA sequencing, the genus Perkinsus presently encompasses seven formally recognized species: P. marinus, P. olseni, P. qugwadi, P. cheasapeaki, P. mediterraneus, P. honshuensis, and P. beihaiensis (Villalba et al. 2004; Moss et al. 2008). Within the genus Perkinsus, P. marinus and P. olseni are recognized as the most detrimental species, known for their significant impact. Diseases stemming from infections by these species are collectively referred to as perkinsosis (Villalba et al. 2004). Perkinsus olseni, formerly recognized as P. atlanticus, is a parasitic organism infecting various marine bivalve species, and it has been documented as a pathogen of Manila clams in regions extending from Europe to Asia, including Korea (Choi and Park 1997; Soudant et al. 2013). Moreover, P. olseni has been observed to parasitize additional clam species, specifically Jedo Venus clam L. jedoensis and the blood cockle Anadarakagoshimensis and Tegillarca granosa within the Korean coastal environment (Park et al. 2006; Cho et al. 2022, 2023). However, it is worth noting that its prevalence and infection intensity appear to be of lesser significance in these clam species compared to the impact observed in Manila clams, emphasizing the diversity of host interactions in the context of P. olseni infections (Park and Choi 2001; Park et al. 2006; Cho et al. 2022, 2023).
This study surveyed parasites in Jedo Venus clams using conventional histological techniques and diagnosis of P. olseni infection using RFTM assay. Our investigation encompassed a comparative analysis of the infection intensity and prevalence of P. olseni within Jedo Venus and Manila clams from similar study sites. Through this comprehensive approach, we aimed to gain insights into the dynamics of Perkinsus infections in these bivalve species, shedding light on the potential variations in parasitic prevalence and intensity across different host clams in the surveyed areas.
2. Materials and Methods
Sampling sites and sample collection
The south coast of Korea is characterized by diverse small bays featuring muddy-sand intertidal and subtidal zones, which serve as habitats for various commercially valuable shellfish species, including the Jedo Venus clam. In September 2022, a total of 200 clams, with SL ranging from 29 mm to 50 mm, were collected from the subtidal flat located in Gamakman Bay, Yeosu, on the south coast (Fig. 1). Upon collection, the shell length was recorded. Subsequently, the soft body was extracted from the shell, and its wet weight was measured. The condition index (CI) of each clam was then computed as the ratio of the dry tissue weight to the respective dry shell weight (CI = (dry tissue weight/dry shell weight) × 1,000).
Histology
For histology, a 3–5 mm-thick section, including the gills, digestive tubule, gonads, and foot was cut from the middle of the clam body. Subsequently, the tissue section was fixed in Davidson's fixative over 24 hours. Following fixation, the specimens underwent a dehydration process using a progressive series of ethanol. Following dehydration, the tissue specimens underwent paraffin embedding, and sections with a thickness of 5 µm were then prepared from the resulting paraffin blocks. Subsequently, these sections were mounted onto glass slides and underwent a staining procedure, which involved the application of Harris’ hematoxylin for primary staining, followed by eosin Y for counterstaining, thus enabling detailed microscopic examination. The clam sections embedded in the histology slides were examined finally under a light microscope, and types of pathogenic organisms were identified.
P. olseni Infection Intensity
Another part of the gill tissue was excised from each clam and placed in 15 ml conical tubes containing 5 ml of Ray's fluid thioglycollate medium (RFTM, Ray 1966), which was fortified with antibiotics, nystatin (200 units/ml) and chloramphenicol (100 ng/ml) to prevent bacterial contamination. Then, the gill tissues underwent a one-week incubation in darkness at room temperature. After the incubation, the gill tissues were digested in 2 M sodium hydroxide (NaOH) at 60°C (Choi et al. 1989). The 2 M NaOH solution was removed by thoroughly rinsing the samples with filtered seawater. Subsequently, the P. olseni hypnospores retained in the tubes were resuspended in a phosphate-buffered saline (PBS) solution. The number of P. olseni hypnospores in a representative subsample was determined using a hemocytometer. The resulting measure of infection intensity was reported as the count of P. olseni cells per gram of gill tissue.
3. Results and Discussion
A total of 200 clams with an average SL of 42.4±3.2 mm were collected and analyzed for histology and RFTM. The clams displayed a mean condition index (CI) of 69. Notably, L. jedoensis clams tend to have denser and heavier shells than Manila clams, likely contributing to the lower CI observed in these individuals.
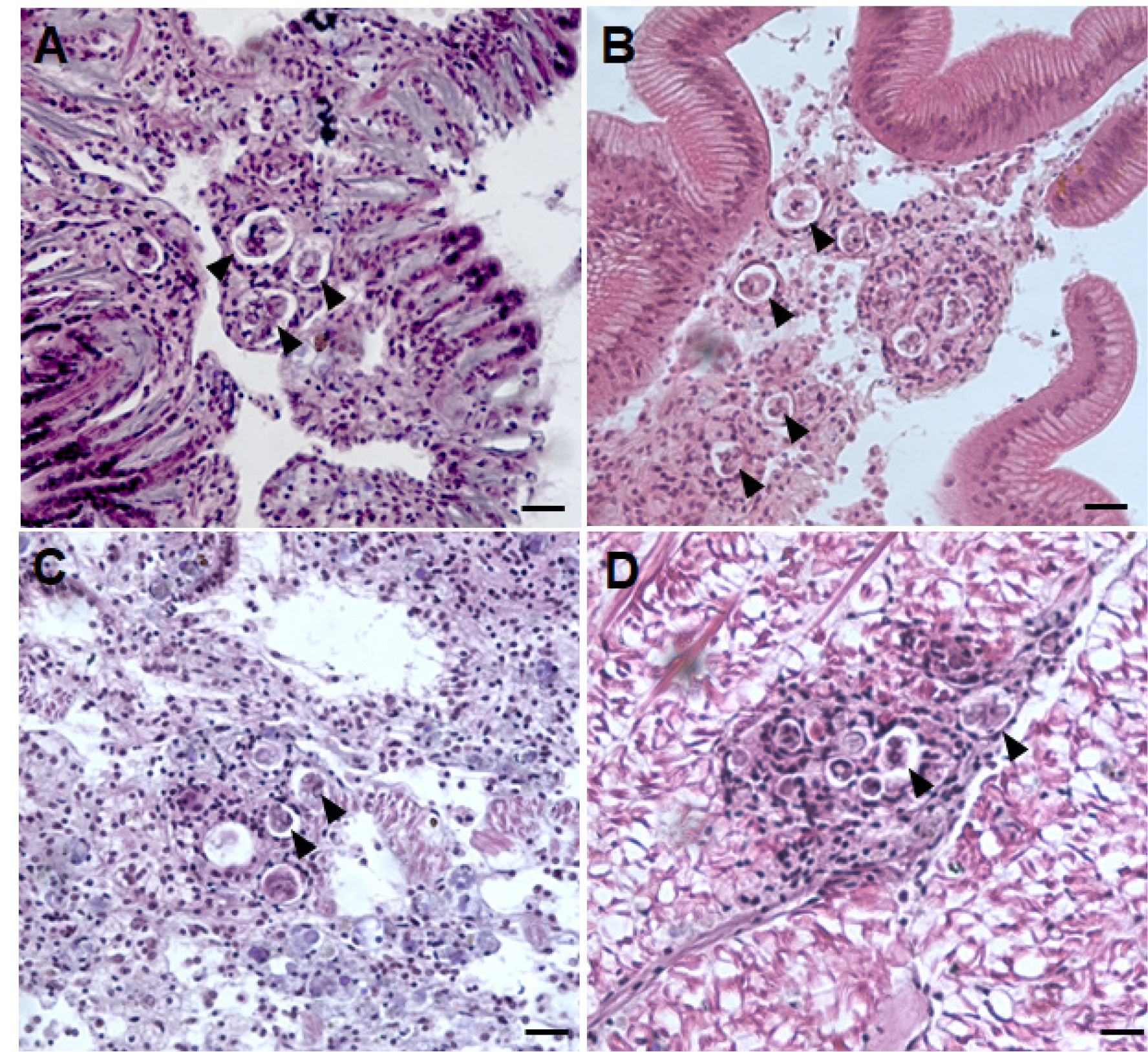
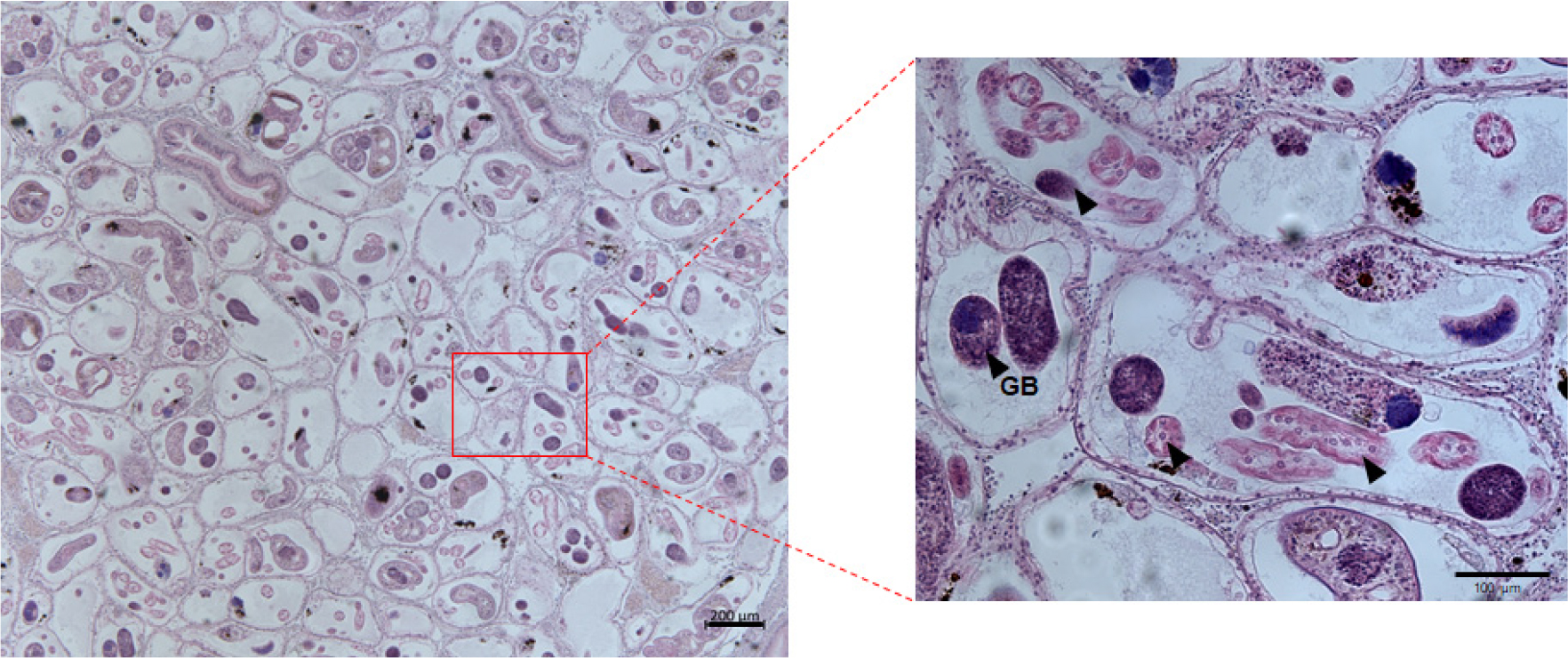
In histology, P. olseni trophozoites could be observed in various regions, including the gills, around the digestive tubules, gonads, and the foot (Fig. 2). These Perkinsus trophozoites displayed characteristic features, including eccentric vacuole and a signet ring appearance, accompanied by marked hemocyte infiltration (Park et al. 2006). Besides the presence of P. olseni, a few clams exhibited larval trematodes and sporocysts containing developing germinal balls within the gonad, as depicted in Fig. 3. The prevalence, the percentage of the infected clams in the total clams examined, of P. olseni and the larval trematode was 1.5% and 1%, respectively.
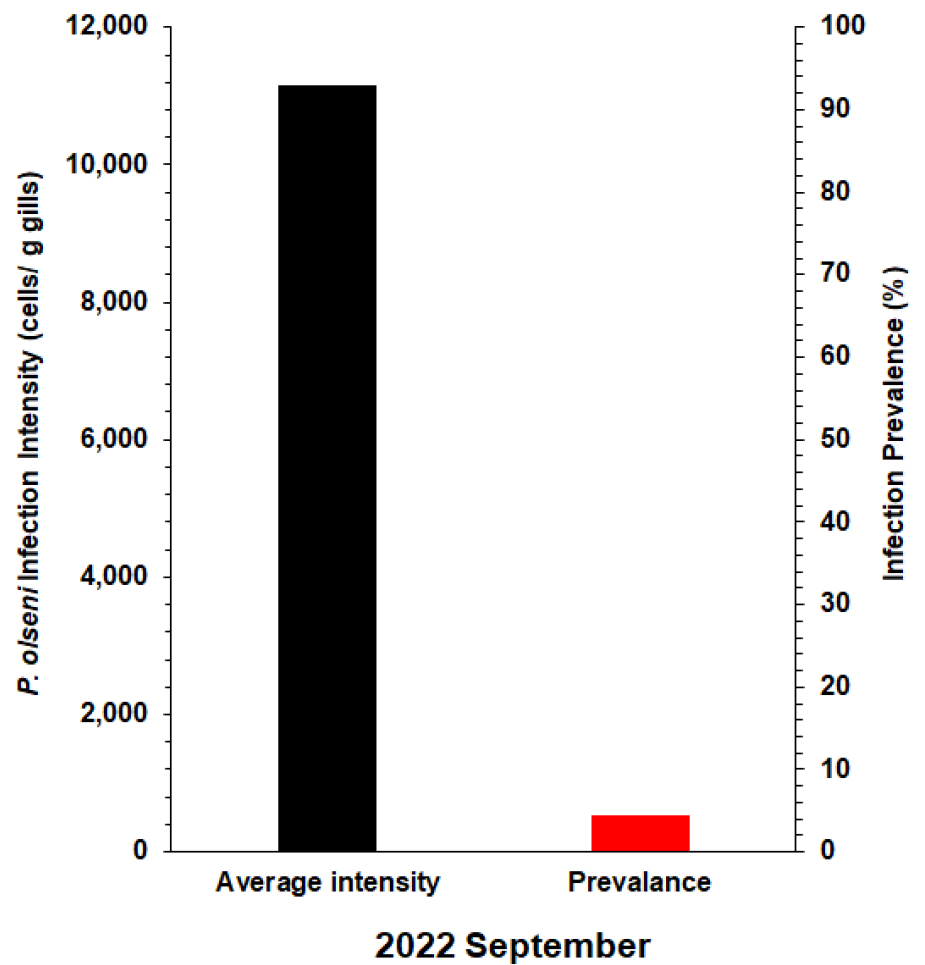
Fig. 4 illustrates the prevalence and mean infection intensities of P. olseni in L. jedoensis determined using RFTM. Consequently, the infection intensity of P. olseni in Jedo Venus clams ranged from 7.0 × 104 to 1.1 × 106, with a mean intensity of 1.1 × 104. A prior study conducted on L. jedoensis (=Protothaca jedoensis) from Yeosu reported varying infection intensities of P. olseni, ranging from 218 to 1.0 × 105 with a mean of 1.1 × 104 in June 2003, and from 356 to 3.1 × 105 with a mean of 0.8 × 104Perkinsus cells per gram of tissue weight in May 2004 (Park et al. 2006). These findings imply that P. olseni may not be a prominent protozoan parasite affecting Jedo Venus clams compared to Manila clams (Table 1), which are heavily infected and impacted by P. olseni in the context of Korea (Lee et al. 2020, 2021; Yang et al. 2021).
Table 1.
A summary of Perkinsus infection intensity and prevalence in bivalves along the southern coast of Korea. (NA- Not available)
|
Location on the South Coast | Host |
Sampling Period | P. olseni Infection Intensity |
Prevalence (%) | Reference |
| Yeosu | Leucoma jedoensis | September | 1.1 × 104 | 4.5 | This study |
| Yeosu | Leucoma jedoensis | May | 0.8 × 104 | 53.9 | Park et al. 2006 |
| Yeosu | Leucoma jedoensis | June | 1.1 × 104 | 37 | |
| Kangjin | Anadara kagoshimensis | September | 1.2 × 105 ± 2.5 × 105 | 13.5 | Cho et al. 2022 |
| Yeoja | Tegillarca granosa | October | NA | 1.4 | Cho et al. 2023 |
| Yeoja | Tegillarca granosa | November | NA | 1.1 | |
| Kangjin | Ruditapes philippinarum | June | 6.9 × 105 ± 1.1 × 106 | 100 | Park and Choi 2001 |
| Wando | Ruditapes philippinarum | June | 1.2 × 105 ± 1.5 × 104 | 84 | |
| Changheong | Ruditapes philippinarum | November | 1.1 × 104 ± 1.0 × 104 | 95 | |
| Yeosu | Ruditapes philippinarum | October | 5.1 × 105 ± 5.1 × 105 | 100 | |
| Mokpo | Ruditapes philippinarum | June | 7.1 × 105 ±3.4 × 105 | 100 | |
| Tongyeong | Ruditapes philippinarum | March | 4.7 × 105 ± 1.4 × 105 | 66 | |
| Geoje | Ruditapes philippinarum | March | 8.7 × 105 ± 8.4 × 105 | 93 | |
| Sachon | Ruditapes philippinarum | March | 2.5 × 105 ± 2.5× 105 | 93 | |
| Jinhae | Ruditapes philippinarum | March | 6.4 × 104 ± 8.0 × 104 | 86 | |
| Wando | Ruditapes philippinarum | May | 3.6 × 106 ± 2.6 × 105 | NA | Kang et al. 2015 |
| Geoje | Ruditapes philippinarum | September | 1.3 × 106 ± 1.2 × 106 | 100 | Lee et al. 2020 |
In our current survey, the prevalence of Perkinsus determined by RFTM was 4.5%, indicating a relatively low occurrence (Fig. 4). However, in a previous study, the reported prevalence was notably higher, with values of 37% for June 2003 and 53.9% for May 2004 (Park et al. 2006). In our study, P. olseni infections were assessed in September, while a previous study conducted the analysis in May and June. Due to this difference in seasons, direct comparisons of infection levels were not feasible. Nevertheless, our findings indicate a decrease in the levels of P. olseni infection in Jedo Venus clams compared to the previous study, suggesting a potential temporal variation in the prevalence of this protozoan parasite in the examined clam population. It is noteworthy that, despite these fluctuations, the prevalence of Perkinsus in Jedo Venus clams remains considerably lower than that reported for Manila clams on the southern coast of Korea (Table 1). In general, Manila clams demonstrate peak P. olseni infection levels, including prevalence and intensity, in September and October during the post-spawning period. Assuming a similar annual infection pattern in Jedo Venus clams, lower infection levels would be anticipated in May and June 2022. However, the observed decrease in P. olseni infection in Jedo Venus clams during this period remains unexplained, potentially attributed to environmental factors or changes in the host, such as the development of resistance against P. olseni. Further investigation is necessary to elucidate the underlying causes of this observed shift in infection dynamics.
The influence of host species on the virulence of P. olseni remains uncertain despite reports of differing susceptibility to P. olseni among various host clam species (Park et al. 2006). Rodriguez et al. (1994) found that P. olseni (=atlanticus) appeared to spread more readily within Ruditapes philippinarum compared to R. decussatus and Venerupis pullastra, as observed six weeks after inoculation with P. olseni zoospores. Since the initial report of P. olseni in Korea (Choi and Park 1997), our comprehension of the sublethal effects of P. olseni infection and its prevalence in Manila clams has expanded substantially, while limited cases reported P. olseni infection and prevalence in other bivalve species. However, Park et al. (2006) previously documented P. olseni infection in Jedo Venus clam, and Cho et al. (2022, 2023) reported the first case of P. olseni infection in the blood cockle A. kagoshimensis and Tegillarca granosa on the south coast of Korea. Despite these findings, there remains a notable absence of information concerning the sublethal consequences of P. olseni infection in a broader array of bivalves beyond Manila clams in the same geographic vicinity, suggesting that host specificity might play a role in the virulence dynamics of P. olseni.
In this study, we observed trematode sporocysts completely occupying the gonad of the Jedo Venus clam. While such infections by trematode sporocysts have been documented in Manila clams residing in fine sediments within shallow to intertidal zones across temperate regions globally, the Northwest Pacific exhibits explicitly at least three morphologically identified species of sporocysts, including Cercaria tapidis, C. pectinata, and Parvatrema duboisi (Ngo and Choi 2004; Park et al. 2008; Le et al. 2015; Jung et al. 2021; Cho et al. 2022). These sporocysts, varying in number and size, contribute to the deterioration of host tissues, particularly in the gonad, a phenomenon recognized as parasitic castration (Baudoin 1975). Although previous studies have reported trematode infections and their consequences on various commercial marine bivalves, the specific species responsible for the observed sporocyst occupation in Jedo Venus clams remains unidentified in this study. Future research efforts are imperative for a detailed understanding of these sporocysts and their potential impacts on the host.