1. Introduction
2. Materials and Methods
Sample collection and strain culture establishment
Morphological observation
DNA extraction, PCR, sequencing, and phylogenetic analysis
DA toxin analysis
3. Results
Morphology of Pseudo-nitzschia lundholmiae, strain PLSG02F8
Molecular analysis
Toxin analysis
4. Discussion
Morphological characteristics
Key to species for Pseudo-nitzschia lundholmiae within Pseudo-nitzschia delicatissima group
Molecular phylogeny and genetic-geographic relationships
Toxin production and implications
Conclusion and further works
1. Introduction
The genus Pseudo-nitzschia includes numerous planktonic diatoms widely distributed across global oceans, with several species known to produce domoic acid (DA), a neurotoxin responsible for Amnesic Shellfish Poisoning (ASP) (Bates et al. 1989; Hasle 2002). DA accumulation through the marine food web poses significant risks to public health and marine ecosystems, and the increasing frequency of harmful algal blooms (HABs) worldwide has heightened concerns regarding toxic Pseudo-nitzschia species (Bates et al. 2018; Dong et al. 2020). Despite their ecological and economic importance, regional studies remain uneven, particularly in East Asian waters (Hasle 2004; Lelong et al. 2012).
Taxonomic studies over the past decades have revealed rapidly expanding species diversity within the genus. The number of recognized Pseudo-nitzschia species increased from approximately 20 in the mid-1990s (Hasle 2004) to 37 by 2012 (Lelong et al. 2012), with recent assessments suggesting over 60 described species (Bates et al. 2018; Guiry and Guiry 2025; Lundholm et al. 2024). Advances in morphological and molecular taxonomy have led to the description of numerous new species, including approximately 18 newly characterized taxa within the past decade (Chen et al. 2021; Gai et al. 2018; Huang et al. 2019; Percopo et al. 2016; Percopo et al. 2022; Teng et al. 2016). However, ecological and toxicological information for many of these species remains limited. Among recently described species, Pseudo-nitzschia lundholmiae has been reported only sporadically from tropical and temperate regions, including Malaysia and the Taiwan Strait (Dong et al. 2020; Lim et al. 2013), and remains poorly studied in terms of morphology, phylogeny, and toxin production. Notably, no confirmed records or detailed characterizations of this species have been reported from Korean waters, leaving its presence and potential ecological impact unresolved.
In South Korea, research on HAB-forming Pseudo-nitzschia species has primarily focused on a limited number of known toxic taxa, and information regarding species diversity, toxicity, and ecological roles remains insufficient (Kim et al. 2015). This knowledge gap is especially pronounced in the coastal waters of Jeju Island, a region characterized by high biodiversity and active fisheries, where HAB events occur regularly but the contribution of toxic Pseudo-nitzschia species has not been clearly identified Lee et al. (2023).
In this study, we report P. lundholmiae from Jeju Island for the first time, based on the successful establishment of a cultured strain. We conducted comprehensive morphological observations and molecular phylogenetic analyses to clarify its taxonomic identity and biogeographic relevance (Ajani et al. 2016; Chen et al. 2021; Percopo et al. 2016; Teng et al. 2016). Furthermore, we assessed its DA production to evaluate its toxicological potential in local marine environments (Dong et al. 2020; Lim et al. 2013). The findings of this study provide the first evidence of P. lundholmiae occurrence in South Korea and offer critical baseline information for HAB monitoring and risk assessment in Jeju coastal waters.
2. Materials and Methods
Sample collection and strain culture establishment
Seawater samples for strain isolation were collected on 7 April 2022 from the coastal waters of Seogwipo (33°13.80'N, 126°34.70'E), Jeju Island, using the research vessel Jira operated by Jeju National University. At each site, surface seawater (~1 m depth) was collected using a 20 µm mesh phytoplankton net. At the time of sampling (7 April 2022), in situ environmental conditions at the collection site were recorded as 16.0°C for surface temperature and 35.0 psu for salinity.
Single cells or single chains of Pseudo-nitzschia were isolated under an inverted microscope (LM; Axioplan, Cal Zeiss, Overkochen, Germany) at 200× magnification. The isolated cells were washed three times with sterile-filtered seawater to remove contaminants and then transferred into individual wells of a 96-well culture plate (Greiner Bio-One GmbH, Frickenhausen, Germany) containing 200 µL of sterile F/2 medium (Guillard and Ryther 1962). For clonal culture establishment, actively growing isolates were transferred into sterile culture flasks (SPL, Daegu, Korea) containing 25 mL of F/2 medium. All cultures were maintained at 18°C under a 12:12 h light:dark photoperiod, with an illumination intensity of 30–50 µmol photons m-2 s-1 provided by cool-white fluorescent lamps. Cells from the stationary growth phase (first-generation cultures) were preserved with Lugol’s solution (Throndsen 1978) for morphological analysis, and pelleted cells were stored at 4°C in the dark for genomic DNA extraction.
Morphological observation
Preserved cells fixed with acidic Lugol’s solution were examined under a light microscope (LM; Axioplan, Carl Zeiss, Oberkochen, Germany) at 400× magnification. and micrographs were taken with digital camera (AxioCam ERc 5c, Cal Zeiss, Overkochen, Germany). The cell sizes of length and width were measured (n = 20) by taken LM images.
For scanning electron microscopy (SEM), the cells were treated with 10% HCl for decalcification and removal of organic debris, followed by rinsing three times with distilled water to remove residual acid. The cleaned samples were then transferred to 100% ethanol, and subsequently dried using a critical point dryer (EMS 3000, Electron Microscopy Sciences, Hatfield, USA). Prior to SEM analysis, samples were sputter-coated with a thin platinum layer (~10 nm) using a Q150R coater (Quorum, Laughton, UK) to improve surface conductivity. SEM observations were conducted using a field emission scanning electron microscope (MIRA 3, TESCAN, Brno, Czech Republic) at an accelerating voltage of 5 kV. Detailed morphological characters including valve shape, striae structure, fibula spacing, and presence and orientation of poroids were recorded and compared with existing descriptions of Pseudo-nitzschia species.
DNA extraction, PCR, sequencing, and phylogenetic analysis
For genomic DNA extraction, cultures of each clonal strain were harvested in 15 mL conical tubes (SPL, Pocheon, Korea) and centrifuged at 10,000 × g for 5 min at 4°C to obtain cell pellets. Genomic DNA was extracted using the DNeasy Plant Mini Kit (Qiagen, Hilden, Germany) following the manufacturer’s instructions, and purified DNA was stored at -20°C until PCR amplification.
The ITS rDNA region was targeted for amplification. Two primer sets were used: UniITSF (GAA GTC GTA ACA AGG TTT CCG) / UniITSR (TTA CGT ATC GCA TTT CGC TG) for ITS1 and PnITSF (ACT TTC AGC GGT GGA TGT CTA) / PnITSR (CTT GAT CTG AGA TCC GGA ATT) for ITS2 (Kim et al. 2015). PCR amplification was conducted using ProFi Taq DNA Polymerase (Bioneer, Daejeon, Korea) in a final reaction volume of 20 µL with the following cycling parameters: initial denaturation at 94°C for 5 min, followed by 36 cycles of denaturation at 94°C for 1 min, annealing at 57°C for 1 min, and extension at 72°C for 2 min, with a final extension at 72°C for 10 min. PCR products were verified by electrophoresis on a 1% agarose gel and subsequently sequenced by an external sequencing service (Macrogen Inc., Seoul, Korea).
Raw ITS1–5.8S–ITS2 sequences were edited, aligned, and trimmed using MEGA v7.0 (Kumar et al. 2016). Phylogenetic trees were constructed using the Maximum Likelihood (ML) method under the General Time Reversible (GTR) substitution model with 1,000 bootstrap replicates. Additional Neighbor-Joining (NJ) and Maximum Parsimony (MP) analyses were performed to confirm tree topology consistency.
DA toxin analysis
To assess domoic acid (DA) production in the isolated Pseudo-nitzschia strain, eight 250 mL Erlenmeyer flasks were prepared, each containing 120 mL of culture standardized to an initial concentration of 2,000 cells·mL-1. The cultures were maintained at 18°C under a controlled 12:12 h light:dark cycle, with illumination provided by cool-white fluorescent lamps at an intensity of 48–52 µmol photons·m-2·s-1. Because DA can degrade over time, particularly during extended culture growth (Tammilehto et al. 2015), toxin measurements were primarily conducted during the first two to three weeks after culture establishment to ensure reliable quantification.
Sampling commenced on day 10 of cultivation and continued at four-day intervals. At each sampling event, 80 mL of culture was gently withdrawn from two independent replicate flasks and filtered through a 47 mm GF/F glass-fiber filter to separate the intracellular and dissolved toxin fractions. Both the filtrates (15 mL) and the filters were immediately frozen and stored at -80°C until chemical analysis. To link toxin dynamics with physiological condition, subsampling was intentionally aligned with specific growth phases of the culture: days 10 and 14 corresponded to log phase, day 18 represented the exponential phase, and day 22 reflected the onset of the death (decline) phase. Throughout the experiment, cell abundance was monitored every two days beginning from day 0 to track the progression of the growth curve and confirm the physiological states associated with each toxin sampling point.
Domoic acid was quantified following the analytical procedure described by Wang (Wang et al. 2007) using high-performance liquid chromatography (Agilent 1260 Infinity II, Agilent Technologies, Santa Clara, USA). Separation was performed on a C18 reverse-phase column using an acetonitrile:water (60:40, pH 4.5) mobile phase, and toxin detection was carried out at 242 nm using a diode-array detector.
3. Results
Morphology of Pseudo-nitzschia lundholmiae, strain PLSG02F8
Cells of strain PLSG02F8 measured 68–76 µm in length and 2.2–2.7 µm in width (Table 1). In valve view, cells are narrowly lanceolate and bilaterally symmetrical, forming stepped colonies in which approximately one-quarter to one-fifth of adjacent cells overlap (Fig. 1a). The valve displays a distinct central interspace with a central nodule (Fig. 1b and c). The density of fibulae and striae is 15–18 and 30–34 per 10 µm, respectively (Table 1). Fibulae are irregularly spaced, and each striae consists of a single row of circular poroids, occurring at a density of 3–5 poroids per 1 µm (Fig. 1c and f). The hymenes of individual poroids are usually divided into 1–3 sectors, although 4 sectors were observed infrequently (1.1% of 185 observations; Figs. 1f and 2a). The valvocopula bears band striae at a density of 36–42 per 10 µm, with each hymen divided into 2–5 sectors (Fig. 1e). A rare occurrence of 6 sectors (2.4% of 124 observations) was also recorded (Figs. 1e and 2b). The apical ends of the valve are bluntly rounded (Fig. 1d).
Table 1.
Comparative morphological characteristics and domoic acid (DA) production capacity of the cultured Pseudo-nitzschia lundholmiae strain from Korea (highlighted in gray) and its phylogenetically allied Pseudo-nitzschia species
| Species | Overlap | Valve Shape | Central interspace | Length (µm) | Width (µm) | Fibulae in 10 µm | Striae in 10 µm | Rows of poroids | Poroids in 1 µm | Band Striae in 10 µm | Toxic |
| P. lundholmiae (Korea) | 1/4-1/5 | lanceolate | + |
68-76 (n = 22) |
2.2-2.7 (n = 20) |
15-18 (n = 20) |
30-34 (n = 20) |
1 (n = 26) |
3-5 (n = 20) |
36-42 (n = 15) | yes |
| P. lundholmiae1 | 1/5-1/6 | lanceolate | + | 63–73 | 1.7-2.3 | 16-18 | 28-34 | 1 | 4–6 | 35-40 | N. A |
| P. cuspidata2 | 1/6-1/7 | lanceolate | + | 48-84 | 1.5-2.3 | 20-26 | 39-44 | 1 | 5-7 | 49-53 | yes |
| P. cuspidata3 | N. A | lanceolate | + | 47-53 | 1.4-2.0 | 19-25 | 35-44 | 1 | 4-6 | N. A | N. A |
| P. plurisecta4 | - | Linear to lanceolate | + | 56–60 | 1.5–2.0 | 17–25 | 34–45 | 1 | 4–7 | 45-48.5 | N. A |
| P. fukuyoi1 | 1/6 | Linear to lanceolate | + | 74-81 | 1.5-1.9 | 17-19 | 32-34 | 1 |
2-3 (rarely 1) | 39-47 | N. A |
| P. pseudodelicatissim3 | - | Linear | + | 70-87 | 0.9–1.6 | 20-25 | 36-43 | 2 | 5-6 | 48-55 | no |
N.A: not available; 1Lim et al. 2013; 2Teng et al. 2021; 3Lundholm et al. 2003; 4Orive et al. 2013.

Fig. 1.
Light microscopy (LM) and scanning electron microscopy (SEM) images of Pseudo-nitzschia lundholmiae strain PLSG02F8 from Jeju Island. LM: (a) LM image showing cells forming characteristic stepped chains in valve view. (b) SEM valve view of a single cell. (c) Detail of valve striae showing a single row of poroids, a well-developed central interspace, and a distinct central nodule. (d) Apical region with bluntly rounded apices. (e) Band striae of the valvocopula. (f) Detailed view of poroid hymen exhibiting a perforated structure composed of multiple small openings. Scale bars as indicated. Scale bars: A: 20 µm, B: 10 µm, C: 2 µm, D: 1 µm, E and F: 0.5 µm
Molecular analysis
The ITS1–5.8S–ITS2 rDNA sequences obtained from the Jeju strains PLSG02F2 and PLSG02G7 were deposited in GenBank under accession numbers PP072219 and PQ799840, respectively. BLAST similarity searches showed that both sequences shared > 99.7% identity with previously reported P. lundholmiae strains from Malaysia (PnTb10, PnTb28) and > 99.6% similarity with strains from China (MC4516, MC4218), confirming their taxonomic placement within P. lundholmiae.
Phylogenetic reconstruction using Maximum Likelihood (ML) analysis revealed that the Jeju isolates clustered within Group I, forming a strongly supported monophyletic clade together with P. lundholmiae strains from Malaysia and China (Fig. 3). This node showed high bootstrap support across all three analytical frameworks, with values of 100/100/100 (ML/NJ/MP), indicating strong topological stability and confirming the genetic coherence of P. lundholmiae as a distinct lineage. Although the clade is positioned near closely related taxa such as P. cuspidata, P. plurisecta, P. pseudodelicatissima, and P. fukuyoi, bootstrap support separating these lineages remained high (≥95%), further demonstrating clear phylogenetic boundaries. Notably, the phylogenetic tree also revealed geographical clustering patterns: Malaysian and Chinese isolates grouped tightly with the Jeju strains, whereas other species within Group I formed separate regional sublineages. This suggests that P. lundholmiae may be more widely distributed across Asian coastal waters than previously documented. The strong molecular support distinguishing P. lundholmiae from its nearest neighbors emphasizes the usefulness of ITS markers for resolving species-complex boundaries within the genus Pseudo-nitzschia.
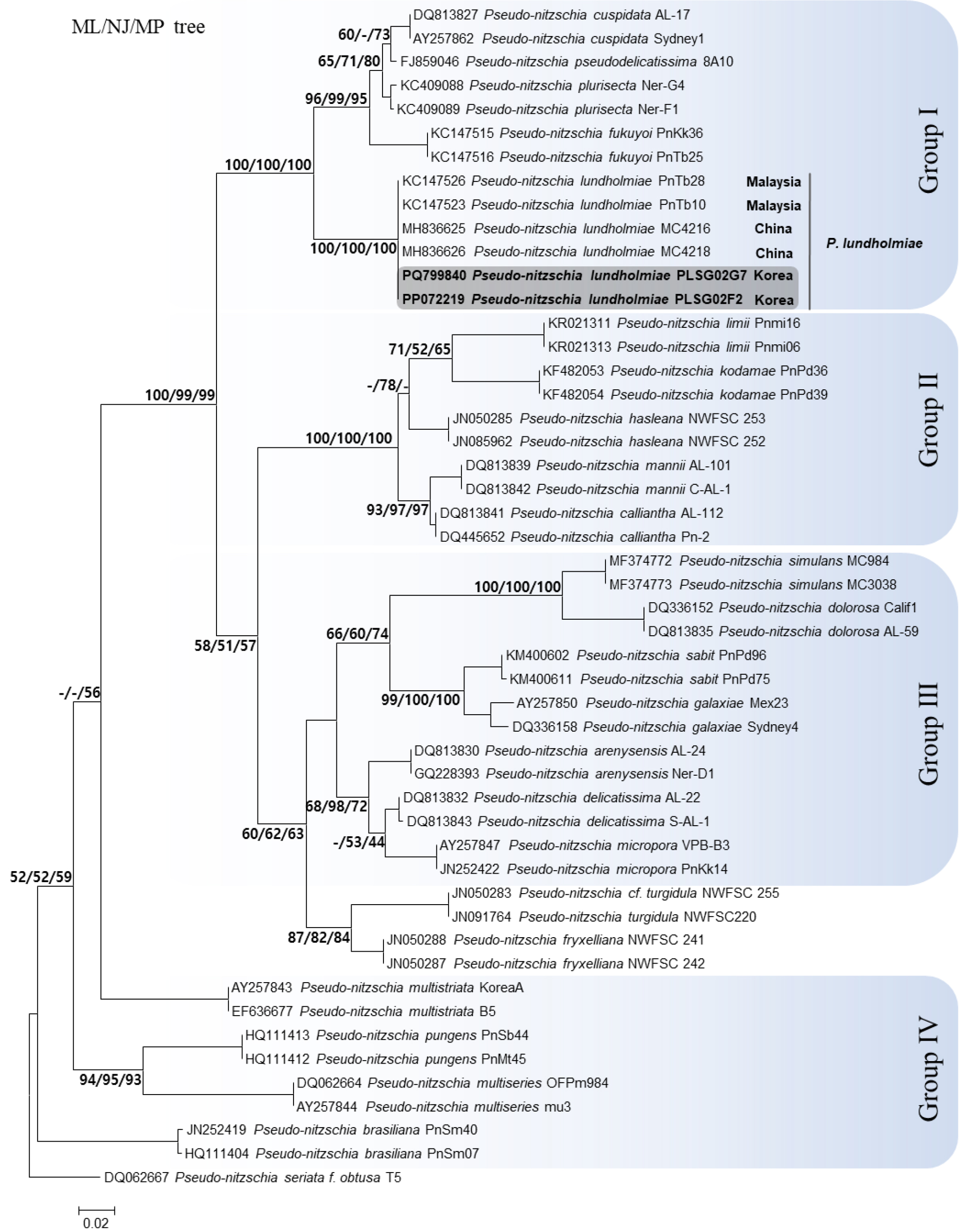
Fig. 3.
Maximum Likelihood (ML) phylogenetic tree inferred from ITS1–5.8S–ITS2 rDNA sequences of Pseudo-nitzschia species. Nodal support values are presented as bootstrap percentages for ML, Neighbor-Joining (NJ), and Maximum Parsimony (MP) analyses (ML/NJ/MP). Values below 50% are not shown. The Korean P. lundholmiae strains (PLSG02F2 and PLSG02G7) are highlighted in gray. Major phylogenetic groups are indicated in blue and labeled as Groups I–IV
Toxin analysis
The temporal changes in cell abundance and intracellular domoic acid (DA) content of the cultured Pseudo-nitzschia lundholmiae strain are shown in Fig. 4. Cell density increased steadily throughout the experiment, reaching a maximum of approximately 1.88 × 105 cells mL-1 on day 18, after which a slight decline was observed by day 22. This pattern reflects a typical batch-culture growth curve, transitioning from exponential growth to stationary and early decline phases.
Intracellular DA was detected at all sampling points; however, toxin yield per cell varied with growth stage. The highest intracellular DA quota was recorded on day 10 at 1.89 fg cell-1, corresponding to the early-log growth phase. From day 10 onward, toxin levels progressively decreased despite continued increases in cell abundance. By day 22, intracellular DA reached its lowest measured value of 0.37 fg cell-1.
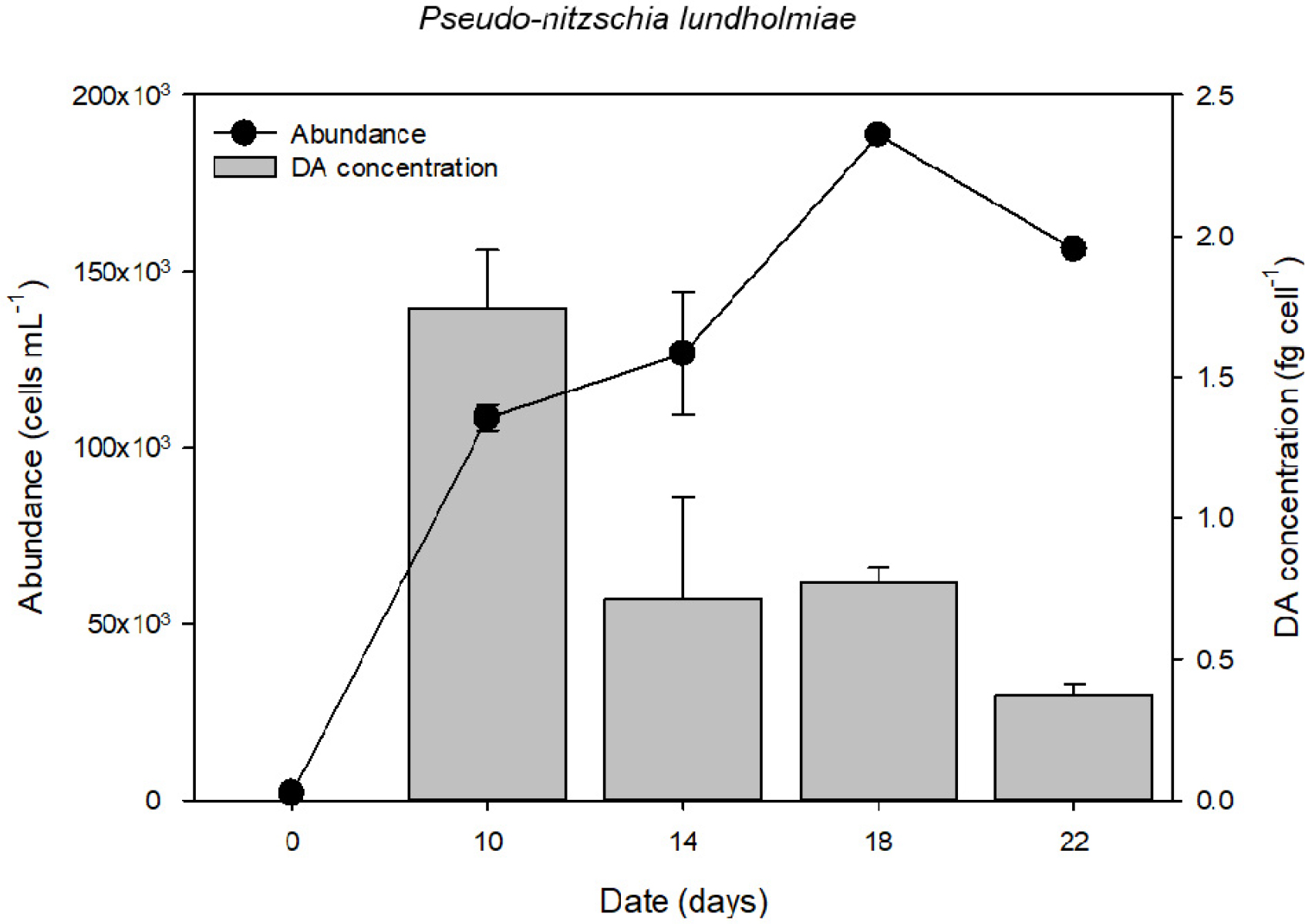
Fig. 4.
Temporal changes in cell abundance and intracellular domoic acid (DA) content of Pseudo-nitzschia lundholmiae strain PLSG02F8 during a 22-day batch culture experiment. Cell abundance is shown as black circles (cells mL-1), and cellular DA content is shown as gray bars (fg cell-1). Error bars represent standard deviation (SD) based on triplicate cultures.
4. Discussion
Morphological characteristics
The morphological characteristics of the Pseudo-nitzschia lundholmiae strains isolated from Jeju Island are largely consistent with the original species description (Lim et al. 2013). Diagnostic features observed in the present study including narrowly lanceolate and bilaterally symmetrical valves, a well-defined central interspace with a central nodule, and broadly rounded apices fall within the known morphological range of the species. Quantitative parameters such as valve dimensions, striae density, fibulae spacing, and poroid arrangement further corroborate the taxonomic assignment.
Based on the diagnostic morphological characters identified in this study, the stepwise keys to species were constructed to distinguish P. lundholmiae from other members of the Pseudo-nitzschia delicatissima group. Within this group, species are first delimited by their narrow cell width (< 3 µm), followed by the presence of a central interspace and the formation of stepped chains. Subsequent separation is achieved using valve symmetry, valve outline, and poroid arrangement. P. lundholmiae is characterized by lanceolate and symmetrical valves with a single row of poroids and, most notably, by a poroid hymen exhibiting a perforated structure composed of multiple small openings. This feature clearly differentiates P. lundholmiae from its closest congener P. cuspidata, whic h possesses poroid hymens subdivided by thin membranous partitions rather than discrete perforations. The inclusion of hymen microarchitecture as a diagnostic character provides enhanced resolution for species discrimination within the morphologically homogeneous P. delicatissima group and highlights the importance of SEM and TEM-based observations for accurate taxonomic identification.
Key to species for Pseudo-nitzschia lundholmiae within Pseudo-nitzschia delicatissima group
Cells are less than 3 µm in width・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・“Pseudo-nitzschia delicatissima group”
1a. Central interspace present・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・2
1b. Central interspace absent・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・other species
2a. Cells forming stepped chains・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・3
2b. Cells solitary / single・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・other species
3a. One row of poroids・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・4
3b. Two rows of poroids・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・other species
3c. Variable rows of poroids・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・P. brasiliana
4a. Symmetrical valve・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・5
4b. Asymmetrical valve・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・P. circumpora
5a. valve lanceolate ・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・6
5b. valve linear to lanceolate・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・other species
6a. Poroid hymen subdivided by thin membranous partitions・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・P. cuspidata
6b. Poroid hymen perforated with multiple small openings・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・・P. lundholmiae
Minor morphological deviations were nevertheless detected. The Jeju strains exhibited slightly increased valve width and a proportionally larger intercellular overlap region compared to previously described populations. Such variation likely reflects intraspecific morphological variation influenced by geographic origin or culture conditions, a phenomenon commonly reported within the genus Pseudo-nitzschia. Although slight differences in valve width and overlap ratio were observed, the relatively large intercellular overlap (1/4–1/5 of cell length) consistently occurred in our cultures and aligns with one of the diagnostic features reported for P. lundholmiae. In Pseudo-nitzschia taxonomy, the degree of cell overlap within stepped chains is considered a stable species-level morphological character distinguishable under LM, although minor variation may occur among strains or under different culture conditions. In particular, variability in hymen sectorization, including the occasional presence of weakly divided or undivided poroids, provides additional ultrastructural resolution that may aid discrimination from closely allied taxa within the P. pseudodelicatissima species complex.
Molecular phylogeny and genetic-geographic relationships
The ITS1–5.8S–ITS2 rDNA phylogeny clearly places the Jeju strains within the P. lundholmiae lineage, exhibiting >99.6% sequence similarity to Malaysian and Chinese strains. Phylogenetic reconstruction revealed that Korean isolates form a strongly supported monophyletic clade with these Southeast Asian strains, with bootstrap support of 100/100/100 across ML, NJ, and MP analyses. Although P. lundholmiae clusters near morphologically similar species such as P. cuspidata, P. plurisecta, P. pseudodelicatissima, and P. fukuyoi, phylogenetic boundaries remain clearly delineated, with high bootstrap separation (≥95%) between lineages. This supports earlier findings that ITS markers provide strong resolution for disentangling cryptic diversity within the genus (e.g. Percopo et al. 2016; Teng et al. 2016). The close clustering of Jeju, Malaysian, and Chinese isolates indicates that P. lundholmiae may indicate potential regional connectivity among currently sampled Asian populations, rather than conclusively demonstrating broad distribution. This reinforces the need for expanded biogeographic surveys to determine whether its distribution reflects natural dispersal, environmental adaptation, or under-detection in previous monitoring programs.
Jeju Island occupies a unique oceanographic position influenced by the Tsushima Warm Current, which transports relatively warm and saline waters from the East China Sea into Korean coastal regions. Although Korean waters are generally classified as temperate, the Jeju region has undergone pronounced subtropicalization in recent decades, with comparatively mild winter temperatures relative to mainland coasts. Such hydrographic characteristics may provide environmental conditions comparable to those reported for P. lundholmiae in tropical and subtropical Asian waters. While direct environmental comparisons remain limited due to insufficient field data from previous studies, the genetic similarity among Asian isolates suggests that oceanic connectivity and regional current systems may suggest the possibility of dispersal and establishment in Jeju coastal waters.
Toxin production and implications
The toxin analysis confirms that the Jeju strain of P. lundholmiae is capable of producing intracellular domoic acid (DA) throughout its growth cycle. The highest per-cell DA quota was observed during early growth (1.89 fg cell-1 on day 10), matching or slightly exceeding previously reported values for this species (Dong et al. 2020). DA concentration declined progressively as cultures approached peak cell density and then entered the decline phase, consistent with patterns observed in other toxic Pseudo-nitzschia species (Tammilehto et al. 2015). These temporal dynamics suggest that DA synthesis in P. lundholmiae may be closely tied to physiological transitions during growth. High DA concentrations during early to mid-log phases may indicate that toxin production is favored under conditions of rapid metabolic activity, whereas dilution, metabolic shifts, or decreased synthesis may contribute to declining toxin quotas in dense or aging cultures. Interestingly, the highest cellular DA quota was detected during the early log phase rather than during stationary growth, which contrasts with the commonly reported pattern of stress-induced toxin enhancement under nutrient limitation in several Pseudo-nitzschia species. In P. multiseries and P. seriata, elevated DA production is frequently associated with silicate or phosphate limitation and late exponential to stationary phases (Bates et al. 1989; Lelong et al. 2012). However, species-specific variability in toxin regulation has been increasingly recognized. For example, Dong et al. (2020) reported measurable DA production by P. lundholmiae under standard nutrient level conditions, suggesting that toxin biosynthesis may not be strictly stress-dependent in this species. The early-log maximum observed in the present study may indicate that DA production in P. lundholmiae is partially coupled to active growth metabolism rather than solely to physiological stress.
When compared to other toxigenic species, the maximum DA quota recorded in the Jeju strain (1.89 fg cell-1) is substantially lower than values commonly reported for highly toxigenic species such as P. australis and P. multiseries, which can exceed tens to hundreds of fg cell-1 under favorable conditions (Bates et al. 2018). Instead, the observed quota falls within the lower range reported for moderately toxigenic members of the P. delicatissima complex. This suggests that while P. lundholmiae may not represent a high-risk bloom-forming toxin producer, its contribution to cumulative DA levels in mixed-species assemblages should not be overlooked.
From an ecological and management perspective, the confirmation of DA production reinforces the need to include P. lundholmiae in harmful algal bloom (HAB) monitoring programs in Korea. Although toxin levels were relatively low compared with highly toxic species such as P. seriata or P. australis, even low concentrations can accumulate through the food web, posing risks to shellfish resources and public health. The spatial overlap between Pseudo-nitzschia assemblages and major aquaculture zones in Korea further underscores the importance of continuous surveillance.
Conclusion and further works
This study provides the first confirmed record of Pseudo-nitzschia lundholmiae in South Korea and offers a comprehensive characterization of its morphology, molecular phylogeny, and domoic acid production. The integration of LM and SEM observations, together with ITS-based phylogenetic evidence, demonstrates that the Jeju strains are morphologically and genetically consistent with previously described P. lundholmiae from Malaysia and China. Moreover, the confirmation of intracellular DA production under culture conditions highlights its toxic potential and its relevance for regional HAB monitoring.
Despite these advances, several key questions remain unanswered. Environmental regulation of DA production in P. lundholmiae is still poorly understood. As toxin levels varied substantially across growth stages in this study, targeted experiments manipulating nutrient availability, light intensity, or temperature are needed to determine the environmental triggers and metabolic constraints that govern DA synthesis. Such work is essential for evaluating the ecological risk posed by P. lundholmiae in natural Korean waters. Further research is also required to elucidate the species’ biogeographical distribution and population structure. The strong genetic similarity among Korean, Malaysian, and Chinese isolates suggests a broader Asian distribution than previously recognized. Whether this reflects natural dispersal, ballast-water transport, or simply a lack of previous detection must be resolved through wider sampling and population genomic approaches. In addition, the ecological role of P. lundholmiae within mixed Pseudo-nitzschia assemblages remains poorly documented. Since multiple species within the genus can co-occur and exhibit varying toxicities, long-term field monitoring incorporating high-resolution molecular tools will be crucial for tracking population dynamics and assessing their combined contributions to HAB events.